论文DOI :10.1038/s41467-026-69779-1全球水资源短缺问题日益严峻,反渗透(RO)技术作为主流的海水淡化手段,其核心聚酰胺复合膜由于缺乏有序的纳米结构和均一的孔径,难以突破渗透性与选择性之间的“权衡效应”。共价有机框架(COFs)因其有序的孔道、均一的孔径和高孔隙率,成为下一代分离膜的理想材料。然而,COF固有的孔径(0.8–5 nm)远大于水合离子的尺寸(~0.72 nm),且其结晶度往往不足,导致其在低压RO海水淡化中的应用面临巨大挑战。现有孔径调控策略(如化学修饰或堆积工程)常因增加位阻而干扰反应动力学,导致结晶度下降,进而影响分离性能。氢键在调控COF结构和功能中发挥着关键作用,通过在框架中引入氢键给体或受体,可增强层内和层间相互作用,从而优化孔道几何结构、提高结构有序性。特别是β-酮烯胺结构,其形成的丰富氢键网络能在保证结构完整性的同时提高结晶度,有望解决COF膜的稳定性与结晶度之间的矛盾。基于此,本文提出一种氢键强化策略,通过将1,3,5-三甲酰基苯(TFB)替换为具有酚羟基的1,3,5-三甲酰基间苯三酚(Tp),使其与苯-1,3,5-三碳酰肼(Bth)反应,形成具有丰富氢键网络的β-酮烯胺结构,从而调控COF的堆积模式(从AA堆积转变为AB堆积),获得更小、更均一的超微孔道(0.64 nm),以实现高效的海水淡化。研究内容:
全球对替代水源的日益依赖凸显了提高脱盐效率的迫切需求。共价有机框架(COFs)凭借其有序的孔隙结构和可调的构型,为下一代脱盐膜展现出巨大潜力。然而,当前的共价有机框架膜常因孔径远超水合单价离子尺寸而无法实现高效的海水脱盐。本文提出一种结构稳定的超微孔共价有机框架膜,用于低压反渗透(RO)脱盐,该膜通过氢键强化策略构建。在醛基邻近引入酚羟基的理性设计,产生了富含氢键的 β -酮烯胺构型,促进了Tp-Bth COF膜中的AB堆叠效应和结晶度提升。所得共价有机框架膜在15巴压力下实现99.6%的氯化钠截留率和1.7L·m-2·h-1·bar-1的水渗透率,展现出高性能低压RO脱盐能力。值得注意的是,这些膜表现出优异的耐酸性,在pH 3的溶液中30天后仍保持初始性能。本研究揭示了一种通过氢键介导的策略,可精准调控共价有机框架的孔道结构以实现高性能脱盐。
Highlight:
氢键强化策略实现COF膜结构精准调控:通过在醛基邻位引入酚羟基,合成了Tp-Bth COF膜,其形成的β-酮烯胺结构赋予材料丰富的层内与层间氢键网络。这一策略有效限制了分子旋转,诱导COF堆积模式从弱氢键的AA堆积(TFB-Bth)转变为高度有序的AB堆积,从而获得更小、更均一的超微孔道(~0.64 nm),解决了传统COF膜孔径过大、结晶度低的难题。
高效的海水淡化性能:优化后的Tp-Bth COF膜在15 bar低压下实现了99.6%的NaCl截留率和1.7 L·m-2·h-1·bar-1的水渗透性,单次运行对硼的去除率达75.9%,综合性能远超现有COF膜和部分商业膜(如BW30)。
机理阐释:结合DFT计算、变温红外和高分辨表征,揭示了氢键网络的三大作用:①增强层间结合能(Tp-Bth为-3.22 eV,远高于TFB-Bth的-1.28 eV),缩小层间距至3.2 Å;②限制分子构象旋转,抑制堆叠缺陷;③平衡溶剂-框架亲和力,促进定向生长。这些协同效应使Tp-Bth COF膜兼具高结晶度、窄孔径分布和优异的选择性,为高性能海水淡化膜的设计提供了新范式。
Fig. 1. Demonstrating hydrogen-bond regulated dual-configurational COF membranes. a, Schematic explanation of the hydrogen-bond-regulated strategy. b, Left panel: Schematic illustration of NaCl rejection by the imine-linked TFB-Bth COF membrane with low rotational energy barriers. Right panel: Schematic illustration of NaCl rejection by the β-ketoenamine-linked Tp-Bth COF membrane with high rotational energy barriers.
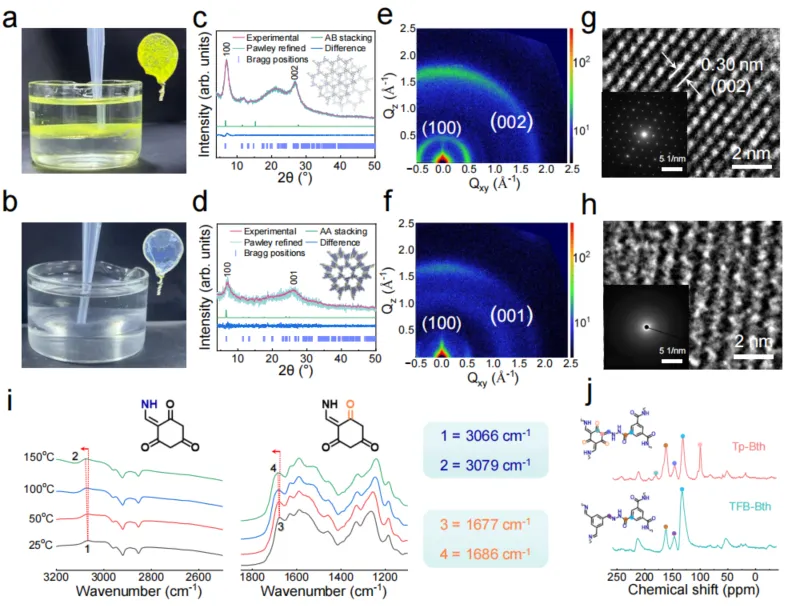
Fig. 2. Structural and functional characterization of free-standing COF nanofilms. The Optical photograph of the freestanding film at the free two-phase interface (Image of the film captured by a wire loop), experimental, Pawley refined, and simulated PXRD profiles of the Tp-Bth film with difference (Rwp = 5.83% and Rp = 4.29%) and the TFB-Bth film with difference (Rwp = 16.74% and Rp = 12.9%), GIWAXS data, and HRTEM image (the lower inset is SAED pattern) of Tp-Bth COF (a, c, e, g) and TFB-Bth COF (b, d, f, h). i, Variable temperature IR spectra of Tp-Bth COF nanofilm; (j) Solid-state 13C NMR spectrum of Tp-Bth COF (top) and TFB-Bth COF (bottom) (400 MHz).
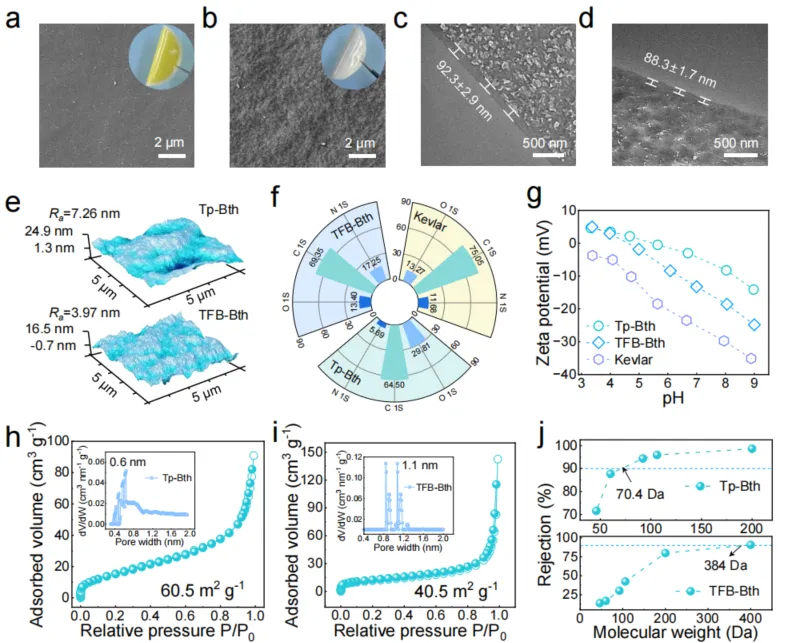
Fig. 3. Morphology characterization and physicochemical properties of the membranes. a-e, Surface SEM, Cross-sectional TEM, and AFM images of Tp-Bth (a, c) and TFB-Bth (b, d) COF membranes (insert: digital images of COF membranes with a diameter of 4.5 cm). f-g, XPS spectra and Zeta potentials of Kevlar, Tp-Bth, and TFB-Bth COF membranes. h-i, N2 adsorption-desorption isotherm and pore size distributions for the Tp-Bth and TFB-Bth nanofilms. j, The rejection performance of uncharged organic solutes (the feed concentration is 1000 ppm).
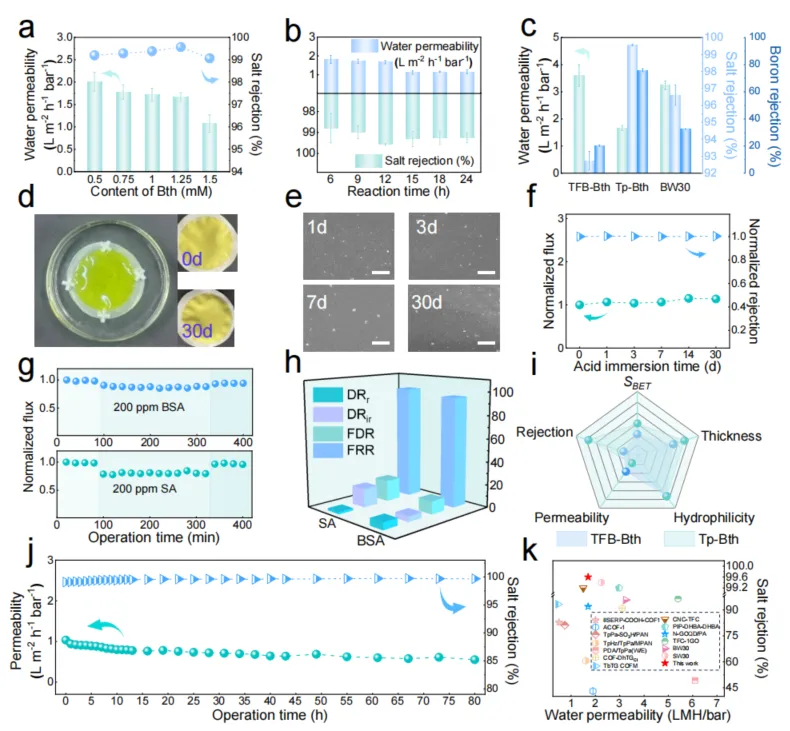
Fig. 4. Performance and application of Tp-Bth and TFB-Bth COF membranes. a-b, Water permeance and NaCl rejection of Tp-Bth COF membranes under different Bth content and reaction time. c, Separation performance of the TFB-Bth, Tp-Bth, and SW30 membranes toward a 2000 ppm NaCl and 5 ppm boron feed solution. d-e, Digital photographs, SEM images (scale bar 1 μm) of Tp-Bth membrane acid immersion process. f, Performance of the long-term pH stability (pH = 3) of the Tp-Bth membrane at a feed pressure of 15 bar and room temperature (2000 ppm NaCl). g, Evaluation of antifouling performance: Normalized water flux versus operation time for Tp-Bth COF membrane using BSA and SA as model foulants. The fouling tests comprise three stages: (1) collecting baseline of water flux using a feed solution of 2,000 ppm NaCl, (2) recording flux when fouled by 200 ppm model foulants in a feed solution of 2,000 ppm NaCl and (3) evaluating flux recovery using a feed solution of 2,000 ppm NaCl after cleaning the membrane via deionized water flushing. h, flux recovery ratio (FRR), flux decline Ratio (FDR), reversible flux decline rate (DRr) and irreversible flux decline rate (DRir) for Tp-Bth COF membrane. i, Radar plot comparing the physicochemical properties of Tp-Bth and TFB-Bth COF membranes. j, Long-term operational stability performance of Tp-Bth membrane. k, Membranes performance in this work compared to other membranes in the literature. Data are presented as mean ± SD (n=3 for Fig. 4a-c).
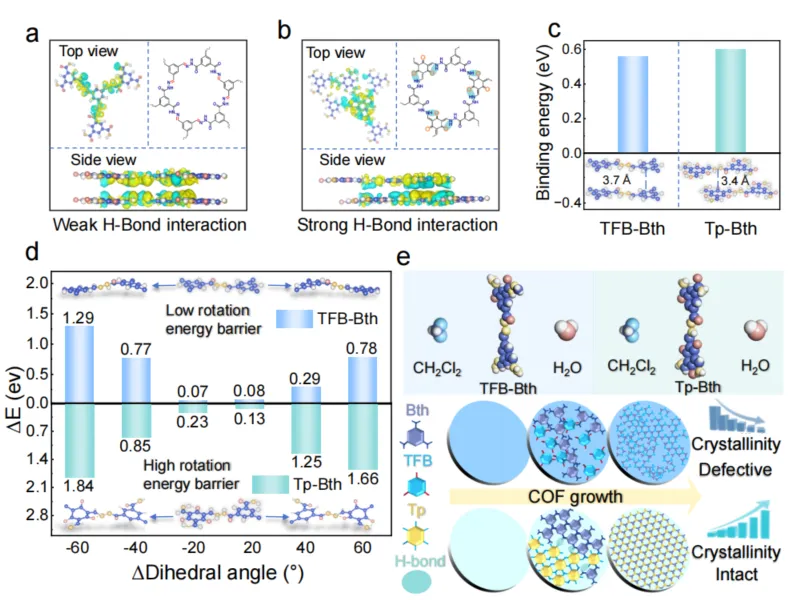
Fig. 5. Mechanistic insights into hydrogen bond formation and performance enhancement. a, Differential charge density of TFB-Bth COF. b, Differential charge density of Tp-Bth COF (blue, decrease in electron density; yellow, increase in electron density). c, DFT calculation of the interlayer binding energy of TFB-Bth COF and Tp-Bth COF. d, DFT analysis of torsional energy barriers in single-layer covalent organic frameworks (COFs), with dihedral angles referenced to the central N-N bond within the molecular framework. Both TFB-Bth and Tp-Bth systems exhibit initial torsion angles of -60 °. The horizontal axis represents the torsional angle variation spanning from -60 ° (minimum) to 60 ° (maximum). The figure illustrates the molecular conformations at dihedral angles of -60 °, 0 °, and 60 °. e, Interaction energy obtained from DFT calculations and the growth of the COF membranes.
结论:
本文成功设计了两种不同的共价有机框架膜,展示了一种氢键调控策略,使Tp-Bth共价有机框架膜具有前所未有的结晶度和卓越的海水淡化性能。这一突破性性能显著超越了最先进的共价有机框架膜。Tp-Bth的特性与氢键的三种协同效应密切相关:通过稳定的 β -酮烯胺构型形成致密的层间和层内氢键网络、诱导高度有序的AB堆叠排列,以及在合成过程中抑制结构缺陷。除了分离能力外,Tp-Bth膜还表现出耐酸性和抗污性能,并具有经过验证的长期稳定性,这对工业应用至关重要。这项工作不仅为设计高度有序的共价有机框架膜提供了一种强有力策略,也为可持续水资源管理的高性能淡化膜确立了基准。
https://doi.org/10.1038/s41467-026-69779-1